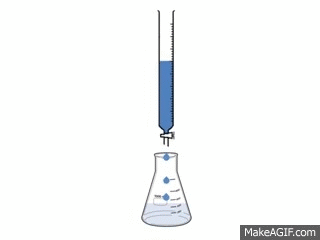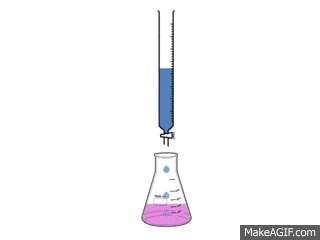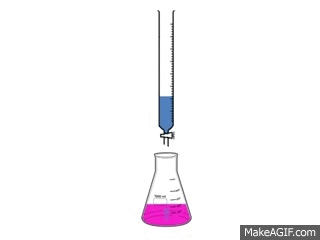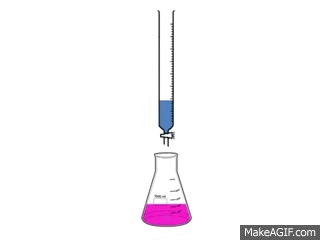
Titrations are one of the simplest, yet most effective ways to find out how acidic or basic a concentration is.
|
|
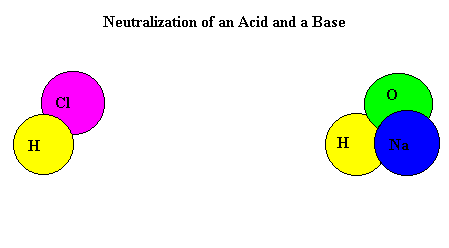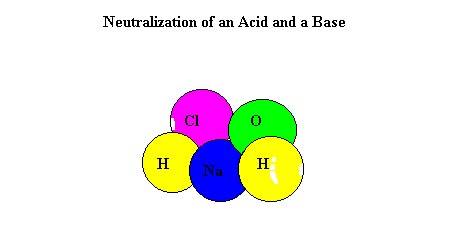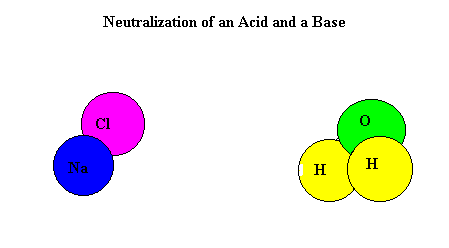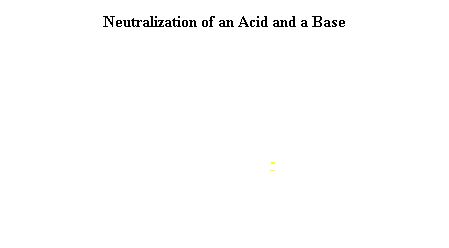
A titration is used to find out how acidic or basic a concentration is by neutralizing it with a different substance. To neutralize a substance, the acid and base needs to be mixed, making the H+ and the OH- separate and form a bond with each other. This creates water and the left over substances create salt.
To make the substance completely neutralize there needs to be an exact amount of acid and base in the substance. The procedure requires: 3-5 drops of Phenolphthalein 50cc of the acid 50cc of the base 1 50 cc buret 1 flask 1 pipet pump 1 10cc pipet The video to the left shows how the reaction looks. |