The general ideas about Acids and Bases.
Properties of Acids and Bases
Acid – Sour, litmus paper turns red, pH below 7, neutralize bases, reacts with metal, H+ donor, starts with H.
Bases – Bitter, litmus paper turns blue, pH above 7, neutralize acids, slippery, reacts with oils, H+ receiver, ends with OH.
|
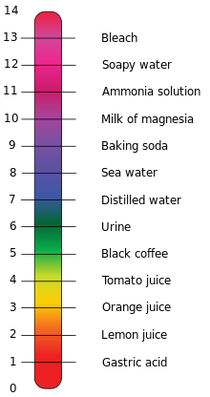
pH scale
The pH scale is used to show how acidic or basic a substance is. Ranging from 0 to 14, it's an easy way of finding the information out. 7+ is a base 7 is neutral 7- is a acid pH indicators (liquid and strips) pH indicators are different ways to measure the pH of an object, usually comparing it by using color to a pH scale. These are different examples of pH indicators. Litmus paper - A more vague way to tell if a substance is acidic or basic, if it is blue it is basic, red it is acidic. pH strips - Used to compare to a colored pH scale, as shown to the left. Universal Indicator - Very similar to pH strips except instead of using strips, the indicator is a liquid. |
How are acids and bases made? |
Nonmetal oxide + water --> Acid
Metal oxide + water --> Base |
Acid/Base Reaction (AKA Neutralization Reaction)
|
Acid + Base --> Salt + Water
HCl+MgOH --> MgCl2+H2O |
Strong acids and strong bases |
A strong acid is an acid that fully dissolves in water, opposed to a regular acid. A strong base does not fully convert into hydroxide ions when put in a a solution, making it fully ionic.
|